
Introduction
Pharmaceutical packaging compliance conversations typically center on labeling and tamper evidence, but strapping — the material that secures and unitizes cases and pallets throughout the supply chain — carries its own compliance weight. A failed strap can mean a failed shipment: product recalls, regulatory holds, and broken cold chain integrity all trace back to load containment failures.
The pharmaceutical industry loses approximately $35 billion annually to cold chain failures, with improper packaging and inadequate sealing cited as primary root causes. When physical load integrity fails, the financial exposure can exceed $500,000 per shipment.
What follows is a regulation-backed compliance checklist for QA teams, packaging engineers, and distribution managers — covering strapping material selection, tension standards, documentation requirements, and FDA/DSCSA alignment from production floor to final delivery.
TLDR
- Pharmaceutical strapping must meet FDA 21 CFR Part 211 and ISTA/ASTM standards — compliance applies to packaging, not just the product inside
- PET strapping works best for heavy pharmaceutical pallets due to superior retained tension; PP suits lighter case loads
- Tension, seal integrity, load configuration, and contamination prevention form the four pillars of compliant strapping operations
- SOPs and batch production records must document all strapping procedures under 21 CFR Part 211.188
- Use the checklist in this guide to avoid the three most common failures: wrong material, under-tension, and missing documentation
Why Strapping Compliance Matters in Pharmaceutical Packaging
The $35B Cold Chain Crisis
Pharmaceutical packaging doesn't end at the primary or secondary container — tertiary packaging (cases, pallets, and bundled units) secured by strapping must maintain product integrity throughout storage, handling, and transport. A failed strap that allows load shift, crush damage, or exposure can result in product rejection, FDA observations, or costly recalls.
A Supply Chain Brain analysis estimates the pharmaceutical industry loses roughly $35 billion globally every year due to temperature excursions, damaged products, and compliance breaches in temperature-controlled logistics. The effectiveness of cold chain monitoring relies heavily on packaging integrity before shipment, with improper packaging and poor sealing identified as primary causes of temperature excursions.
Regulatory Exposure and Downstream Liability
FDA 21 CFR Part 211 Subpart G holds manufacturers responsible for the integrity of packaging operations broadly — including preventing cross-contamination and ensuring materials are suitable for their intended use. Strapping must meet this standard in the context of the full packaging operation, not just at the individual container level.
Beyond regulatory exposure, non-compliant strapping practices create downstream liability:
- Damaged shipments reaching distributors or pharmacies
- Cold-chain breaks for temperature-sensitive drugs
- Documentation gaps that complicate audits
- Rejected loads at distribution centers (up to 11% of unit loads show case damage upon arrival)
Retailers and distributors frequently reject entire unit loads at the dock even if only one or two cartons are damaged, making reliable pallet unitization essential to preventing supply chain delays.
Regulatory Framework Governing Pharmaceutical Package Strapping
21 CFR Part 211 Packaging Control Requirements
Pharmaceutical packaging operations are strictly governed by 21 CFR Part 211 Subpart G, with two sections directly impacting strapping:
21 CFR 211.130 (Packaging and Labeling Operations) requires written procedures designed to assure correct labels, labeling, and packaging materials are used. These procedures must:
- Prevent mix-ups and cross-contamination through physical or spatial separation
- Mandate inspection of packaging facilities immediately before use to assure all drug products and unsuitable materials from previous operations have been removed
- Document inspection results in batch production records
21 CFR 211.122 (Materials Examination and Usage Criteria) requires written procedures covering the receipt, identification, storage, handling, sampling, examination, and testing of packaging materials. Any materials that don't meet written specifications must be rejected before use.
ASTM D4169 vs. ISTA 3A for Distribution Validation
To validate that tertiary packaging can withstand distribution hazards, the industry relies on simulation standards:
ASTM D4169 is an FDA-recognized consensus standard that provides a uniform basis for evaluating shipping units using a sequence of anticipated hazard elements encountered in various distribution cycles. Distribution Cycle 13 (DC 13) is commonly used for small parcels, while DC 6 or DC 12 are used for unitized/palletized loads.
ISTA 3A is a general simulation test for individual packaged-products weighing 70 kg (150 lb) or less shipped via parcel delivery. However, ISTA 3A uses simultaneous top-load vibration, which can cause uncharacteristically severe crushing damage not typically experienced in commercial shipments, making ASTM D4169 the preferred standard for medical and pharmaceutical packaging.
Supporting Standards
ISO 15378 specifies quality management system requirements for primary packaging materials for medicinal products, incorporating GMP principles. While its scope covers primary packaging, its traceability, risk management, and documentation requirements apply in practice to all packaging materials — including strapping used in controlled pharmaceutical environments.
ASTM D3953 covers flat steel strapping and seals intended for use in closing, reinforcing, bundling, unitizing, and palletizing, specifying physical and mechanical property requirements including breaking strength, elongation, and seal joint strength.
Choosing the Right Strapping Material for Pharmaceutical Compliance
Material Performance Comparison
Selecting the correct strapping material is critical for maintaining load integrity without damaging the product.
| Material | Break Strength Range | Elongation & Recovery | Best Pharmaceutical Use Case |
|---|---|---|---|
| Polypropylene (PP) | 120-600 lbs | High elongation, low retained tension | Light/medium duty, compressible loads, short transit |
| Polyester (PET) | 400-4,200 lbs | Low elongation, high retained tension | Heavy/rigid loads, long-term storage, steel replacement |
| Steel | Highest | 1% elongation, poor recovery | Extreme heavy duty (declining in pharma due to risks) |
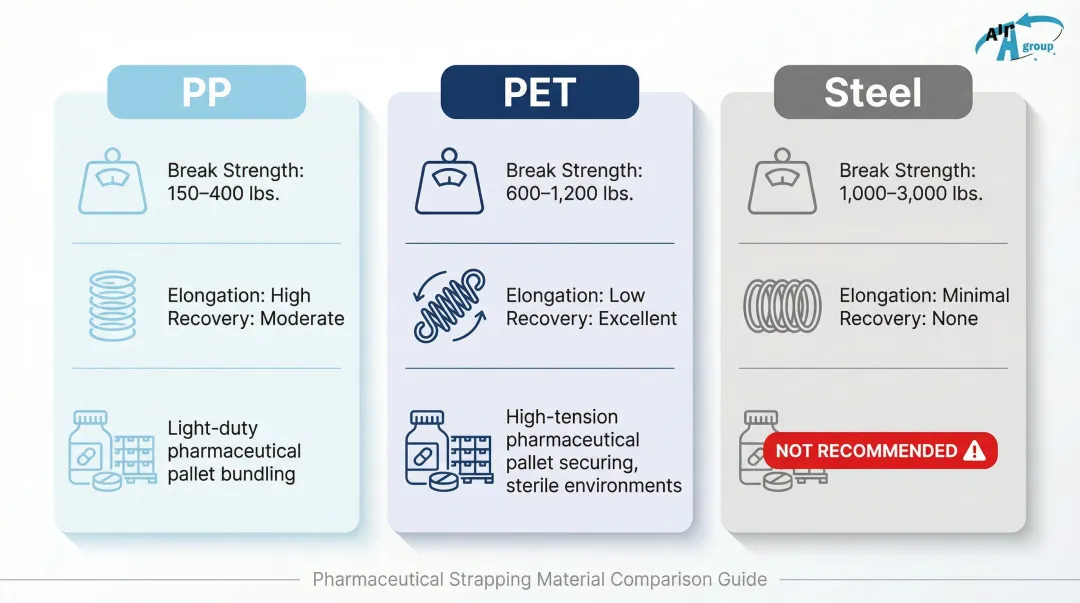
Polypropylene (PP) Strapping
PP strapping is the most widely used option for lighter pharmaceutical case loads. It's cost-effective, available in various break strengths, and poses low contamination risk. However, PP is not ideal for heavy pallets or high-tension applications because it has lower elongation recovery and can loosen over time, creating load instability.
Polyester (PET) Strapping
Where PP falls short, PET picks up. PET strapping is the preferred choice for heavier pharmaceutical pallets and high-tension applications, offering:
- Superior retained tension over extended periods
- Better shock absorption during transit
- Lower risk of load shift
- "Elastic memory" that allows it to stretch under tension and recover after initial application
This elastic memory keeps the strap tight around cooling, shrinking, or settling packs — where steel would simply go slack.
Steel Strapping: Contamination Risks
While high-strength, steel poses significant contamination risks in pharmaceutical environments:
- Metal fragments (0.3 inch to 1 inch in length can cause traumatic injury per FDA guidelines)
- Rust and oxidation
- Safety hazards during handling
UNICEF explicitly states in their packaging specifications that "Steel straps are not acceptable" to prevent contamination during storage and transportation. Most pharmaceutical manufacturers have moved to PET as the functional alternative.
Alliance Packaging Group carries both polypropylene and polyester strapping suitable for pharmaceutical applications, with nationwide delivery that supports consistent supply for packaging and procurement teams.
The Pharmaceutical Strapping Compliance Checklist
Strapping Material Compliance
Verification Requirements:
- Confirm strapping material has been reviewed against written specifications and approved for pharmaceutical packaging environments
- Verify no risk of chemical interaction, contamination (particulates, rust, outgassing), or physical degradation that could compromise product safety
- Ensure incoming strapping materials are inspected upon receipt per 21 CFR Part 211.122
- Maintain records for each shipment indicating receipt, examination, and acceptance or rejection status
Documentation to Maintain:
- Material specifications (type, grade, break strength, width)
- Approved supplier list
- Incoming inspection records
- Certificates of conformance or analysis
Tension and Seal Integrity
Tension Requirements:
Strapping must be applied at the correct tension for the load type: too loose risks load shift and damage; too tight risks crushing of packaging or product.
Recommended Tension Ranges:
- PET strapping should be tensioned to 40-60% (up to 70% for some applications) of its ultimate break strength
- Applying PET at higher tension, rather than using thicker strap, yields higher residual tension
- Tension settings must be validated during equipment qualification
Seal Integrity Testing:
Seals (buckles, heat welds, or friction welds) must be tested for joint efficiency — the ratio of seal strength to strap break strength.
Joint Efficiency Benchmarks:
- Friction welding consistently achieves 75-85% joint efficiency
- Heat welding is effective but subject to temperature-variance defects
- Friction welding eliminates temperature fluctuation risks and is the standard for battery-operated hand tools
Acceptance Criteria:
- Minimum 75% joint efficiency for friction-welded PET/PP straps
- Visual inspection for weld defects (incomplete fusion, charring, misalignment)
- Pull testing at defined intervals per SOP
Load Configuration and Pattern
Configuration Requirements:
The strapping pattern (number of straps, placement, spacing, and direction) must be appropriate for:
- Load weight and dimensions
- Distribution hazards the product will face
- Pallet stacking pattern (columnar vs. interlocked)
Pallet Stability Considerations:
- Columnar stacking gives best overall compressive strength
- Interlocked stacking provides greater lateral strength and stability
- Cross-strapping or edge-protected configurations may be required for pharmaceutical pallets to prevent product movement
Validation Through Distribution Testing:
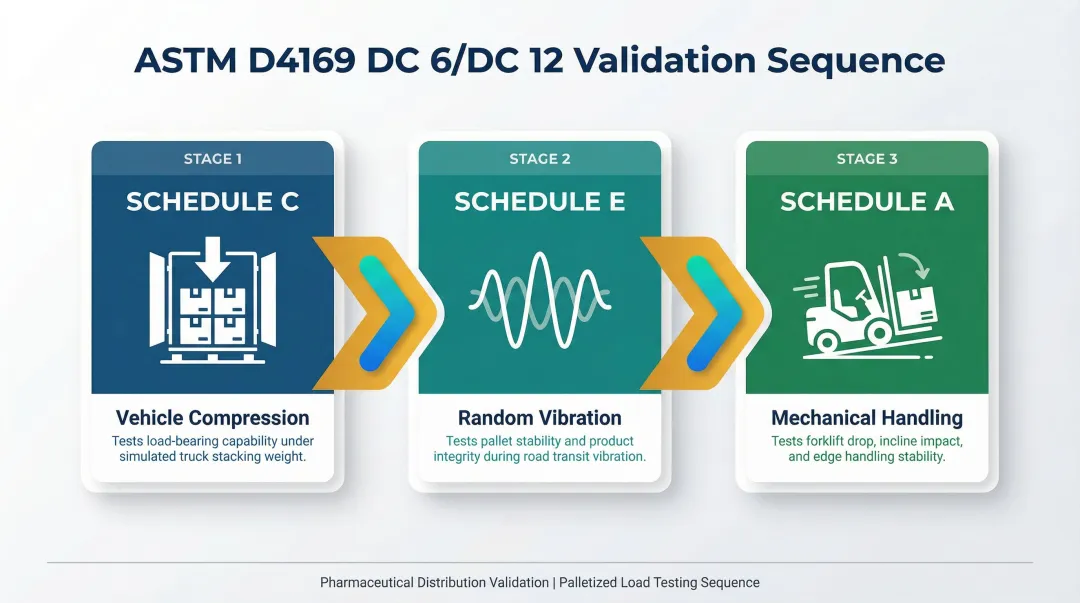
Once your stacking pattern and strap placement are established, the configuration must be defined in the packaging SOP and validated through distribution simulation testing — specifically ASTM D4169 DC 6 or DC 12 for palletized loads — to confirm adequacy for the intended shipping environment.
Test Sequence Includes:
- Schedule C (Vehicle Compression): Tests load-bearing capability
- Schedule E (Random Vibration): Tests stability during truck handling
- Schedule A (Mechanical Handling): Tests stability through forklift obstacle courses and incline impacts
Contamination Prevention During Strapping Operations
Material Storage and Handling:
- Store strapping materials away from potential contaminants in designated clean areas
- Inspect strapping equipment for metal fragments or debris before each production run
- Define procedures for handling and storing strapping materials before use in written SOPs
Line Clearance Requirements:
Per 21 CFR 211.130(e), inspect packaging facilities immediately before use to confirm all drug products and packaging materials unsuitable for subsequent operations have been removed. Results must be recorded in batch production records.
Equipment Sanitation:
Under 21 CFR 211.67(a), all equipment — including strapping machines and palletizers — must be cleaned, maintained, and sanitized at appropriate intervals. This prevents malfunctions or contamination that could alter the safety, identity, strength, quality, or purity of the drug product.
Documentation and Recordkeeping for Strapping Operations
Written Standard Operating Procedures
Strapping specifications must be captured in written SOPs as required by 21 CFR Part 211.130, including:
- Material type and approved suppliers
- Tension settings by load type
- Seal type and joint efficiency requirements
- Strapping pattern (number, placement, spacing)
- Line clearance procedures
- Contamination controls
SOPs must be current, approved, and followed by all personnel involved in packaging operations.
Batch Production Records
21 CFR 211.188 requires batch production and control records include complete information relating to production and control of each batch. For tertiary packaging, this includes:
- Dates of the operation
- Identity of individual major equipment and lines used
- Inspection of the packaging area before and after use
- Identification of persons performing and directly supervising each significant step
Strapping operates at the tertiary packaging level, but specifications still belong in SOPs. Any deviations should be documented through the quality system.

Equipment Calibration and Maintenance
Strapping equipment — tensioners, sealers, and automated strapping machines — must be included in your facility's formal calibration schedule. Under 21 CFR 211.68(a), all mechanical and electronic equipment used in packing must be routinely calibrated, inspected, or checked per a written program, with records maintained. 21 CFR 211.182 extends this to cleaning and maintenance logs.
Records to Maintain:
- Calibration dates and results
- Tension verification tests
- Preventive maintenance activities
- Cleaning and sanitation logs
- Equipment qualification documentation
Deviation Management
Any strapping deviation — wrong material used, seal failures, tension out of spec — must be captured in your deviation management system. Each deviation requires a root cause investigation and documented corrective action, per 21 CFR 211.100(b).
Common Strapping Compliance Mistakes to Avoid
Using the Wrong Strapping Material
A common and costly error is defaulting to whatever strapping is cheapest or most available without verifying it meets written specifications for pharmaceutical use.
High-Risk Scenarios:
- Steel strapping in pharmaceutical environments (contamination risk)
- PP strapping on heavy pallets where PET is required for retained tension
- Material not inspected or approved per 21 CFR Part 211.122
Prevention: Establish approved material specifications in SOPs and conduct incoming inspection before use.
Under-Tensioning Straps to Avoid Product Damage
Well-intentioned operators sometimes reduce strapping tension to avoid damaging lightweight pharmaceutical cartons, but under-tensioned straps allow load movement during transport, which creates a far greater compliance and safety risk than the problem they were trying to prevent.
The Right Approach:
- Correct packaging design (corner protectors, edge boards) should address fragility
- Maintain proper tension per validated specifications
- Don't compromise load security to compensate for inadequate packaging design
Failing to Document Strapping Operations Adequately
Strapping is often treated as an informal step rather than a documented packaging operation. That gap catches up with facilities during audits. Missing SOPs, incoming material records, calibration logs, and deviation reports are exactly what investigators look for.
FDA Warning Letter Evidence:
Recent FDA enforcement actions cite 21 CFR 211.130(e) for failing to adequately inspect packaging lines before use and 21 CFR 211.182 for missing equipment maintenance logs.
In one case, investigators observed capsules and tablets on a blister packing machine during a different product run, despite batch records showing line clearance had been completed — a documentation failure that resulted in a Warning Letter.

Documentation Standard: Integrate strapping into formal packaging SOPs, batch records, and quality systems with the same rigor applied to primary packaging operations.
Frequently Asked Questions
What are the FDA requirements for pharmaceutical packaging?
FDA requirements for pharmaceutical packaging are primarily governed by 21 CFR Part 211 Subpart G (Current Good Manufacturing Practice), which mandates written procedures for all packaging operations, material inspection and approval, labeling controls, and documentation through batch production records.
What is the ISO standard for pharmaceutical packaging?
ISO 15378 is the primary international standard for pharmaceutical packaging materials, applying GMP principles to primary packaging for medicinal products. ISO 11607 covers packaging for terminally sterilized medical devices.
What type of strapping is best for pharmaceutical packaging?
Polyester (PET) strapping is the top choice for pharmaceutical pallets and heavy loads due to its superior retained tension and shock absorption. Polypropylene (PP) works for lighter case-level applications, while steel strapping should be avoided in pharmaceutical environments due to contamination risks.
Does pharmaceutical strapping need to be included in batch production records?
Strapping operates at the tertiary packaging level, but 21 CFR Part 211 still requires written procedures and documentation for all packaging operations. Capture strapping specifications in SOPs and document any deviations through your quality system.
What are the contamination risks of strapping in pharmaceutical packaging?
Steel strapping poses the highest risk, producing metal particulates and rust that can contaminate pharmaceutical products. Polypropylene and polyester strapping carry less risk but still require controlled storage and handling to prevent particulate or chemical contamination.


